Penn State History Lesson: Frank C. Whitmore & Organic Chemistry Carbocation Rearrangements

*This one goes out to the dextrose daddies and maltose mamas STEM nerds*
There’s something oddly satisfying about learning a fundamental concept in class and hearing a professor subtly mention that it’s named after a Penn Stater.
Frank C. Whitmore was the dean of the School of Chemistry and Physics from 1929 to 1947 and the brains behind carbocation rearrangements in organic chemistry. Whitmore Laboratory was named after him due to his research contributions and dedication to Penn State chemistry.
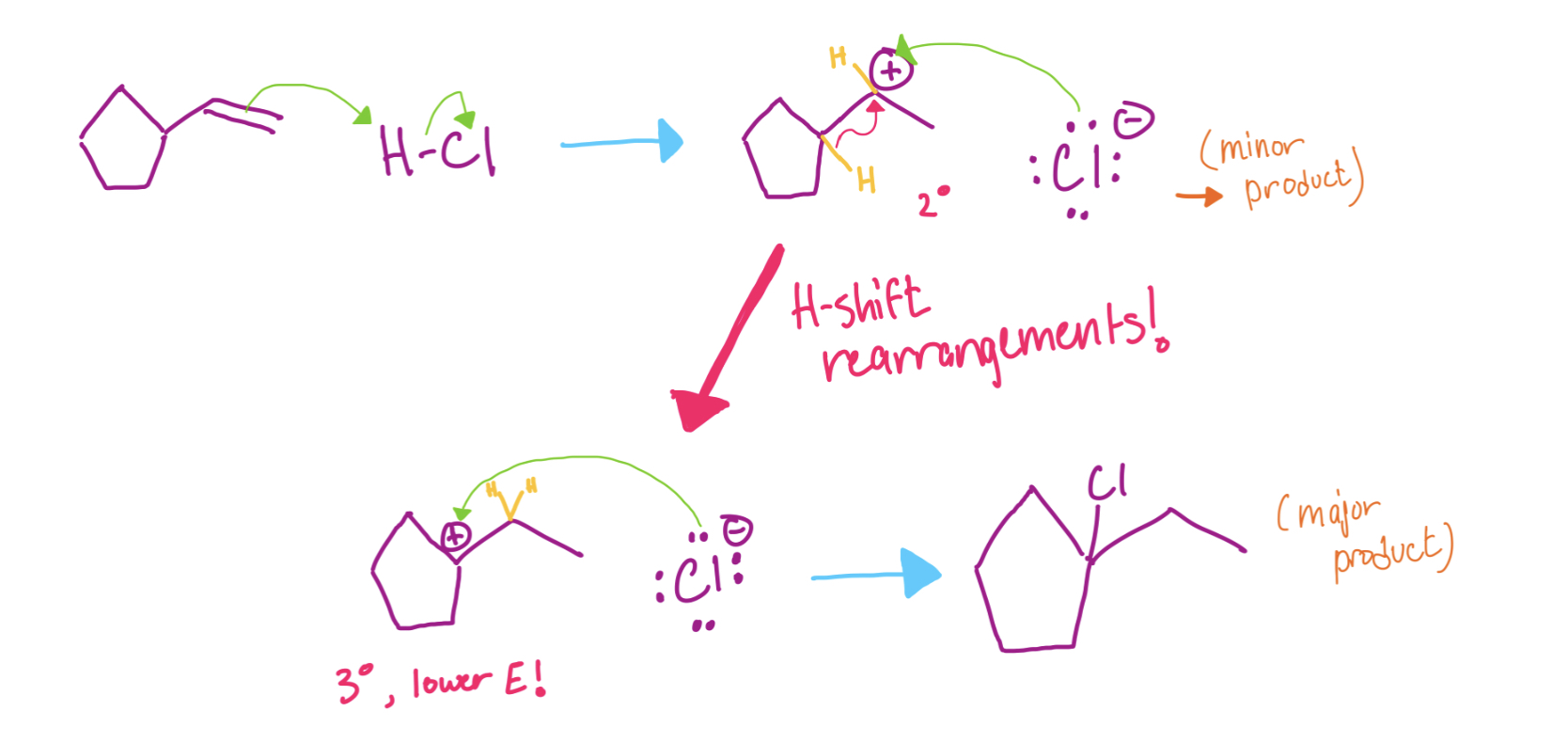
Whitmore shifts, commonly referred to as hydride shifts, is a concept explained in lower-level organic chemistry courses.
Secondary carbocations will rearrange if there is “branching” next door and moving of a group (H or alkyl) results in a more stable carbocation. Because of the increased stability and lower energy, the rearrangement happens very quickly.
Whitmore shifts involve a hydride moving its electrons next door to a carbocation. Because the electrons are moving away from a tertiary site, the result is a carbocation that is more stable at that position.
Whitmore’s research provided evidence for the existence of carbocation rearrangements and published his findings in “The Common Basis of Intramolecular Rearrangements.” Despite criticism, his findings are still the best explanation behind this concept that chemists have today.
Additionally, in 1937, Whitmore published the first advanced organic chemistry textbook written in English, aptly titled “Organic Chemistry.”
Whitmore received his bachelor’s degree and Ph.D. from Harvard in the 1910s and taught at Minnesota and Northwestern before coming to Penn State. Despite taking on a prominent leadership role at the university and recruiting several bright chemists, Whitmore managed to find time to work on his detailed research on carbocation mechanisms.
Whitmore inspired and taught many young Penn State chemists and is often regarded as the person who made Penn State chemistry what it is today.
To learn more about the people and history behind common Penn State building names, check out our comprehensive list.
Your ad blocker is on.
Please choose an option below.
Purchase a Subscription!